
介绍
角膜溃疡是一种严重的情况,其特点是在角膜(眼睛透明的前表面)上出现开放性溃疡。如果未能及时诊断和治疗,它可能导致视力丧失。[1]本指南的目的是为医护专业人员提供与角膜溃疡相关的症状、成因、诊断步骤、可能的干预措施和患者教育的全面概述。
代码
-
- ICPC-2代码:F85 角膜溃疡
[2]
- ICD-10代码:H16.0 角膜溃疡
[3]
症状
-
- 眼红:受影响的眼睛可能呈现红色或血丝状。
[4]
- 眼痛:患者可能在受影响的眼睛中感觉到中度至重度的疼痛。
[4]
- 视力模糊:视力可能变得模糊或朦胧。
[4]
- 对光敏感:患者可能对光的敏感性增加。
[4]
- 眼泪过多:受影响的眼睛可能产生过多的泪水。
[4]
- 眼部分泌物:患者可能有来自受影响眼睛的黄色或绿色分泌物。
[4]
成因
-
- 细菌感染:如金黄色葡萄球菌或铜绿假单胞菌等细菌可导致角膜溃疡。
[5]
- 病毒感染:如单纯疱疹病毒或水痘带状疱疹病毒等病毒可导致角膜溃疡。
[5]
- 真菌感染:如念珠菌或曲霉菌等真菌可导致角膜溃疡。
[5]
- 损伤:角膜损伤,如划伤或异物,可导致角膜溃疡。
[6]
- 隐形眼镜佩戴:不正确的使用或不良卫生习惯可增加角膜溃疡的风险。
[6]
诊断步骤
病史
-
- 收集关于患者风险因素的信息,如隐形眼镜使用、近期眼部创伤或既往眼部感染。
[7]
- 询问症状,包括眼痛、发红、视力模糊和对光的敏感性。
[7]
- 询问任何潜在的健康状况,如糖尿病或免疫抑制,这些可能增加角膜溃疡的风险。
[7]
体格检查
-
- 检查受影响的眼睛是否有发红、肿胀、分泌物或任何可视的角膜异常。
[8]
- 评估视力以确定视力损失的程度。
[8]
- 进行裂隙灯检查,详细检查角膜,识别任何角膜溃疡或浸润。
[8]
实验室检测
-
- 角膜刮片:采集角膜组织样本进行微生物分析以识别致病微生物。
[9]
- 培养和敏感性测试:对角膜刮片进行培养,以确定导致角膜溃疡的特定细菌、病毒或真菌,并评估其抗微生物药物的敏感性。
[9]
诊断成像
-
- 荧光素染色:应用荧光素染料于眼部以识别和可视化角膜溃疡或缺损。
[8]
- 前段光学相干断层扫描(OCT):利用OCT成像评估角膜溃疡的深度和范围,并评估底层结构的参与情况。
[8]
其他检查
-
- 眼内压测量:测量眼内压以排除青光眼,这可能与角膜溃疡相关。
[8]
- 血液检测:进行血液检测,如全血细胞计数和炎症标志物,以评估整体健康状况并识别任何潜在的系统性疾病。
[8]
跟进和患者教育
-
- 安排定期的随访预约,以监测角膜溃疡的发展并在必要时调整治疗。
[10]
- 教育患者关于正确的隐形眼镜卫生的重要性以及佩戴隐形眼镜的相关风险。
[10]
- 指导患者避免揉搓或触摸受影响的眼睛,并认真遵循规定的治疗方案。
[10]
- 强调如果症状恶化或出现新的症状,如严重的眼痛或视力丧失,需要及时就医。
[10]
可能的干预措施
传统干预措施
药物:
角膜溃疡的前5种用药:
- 氟喹诺酮眼药水(例如,环丙沙星,氧氟沙星):
- 费用:每瓶$10-$50。
- 禁忌症:对氟喹诺酮类药物过敏。
- 副作用:眼部刺激,灼烧感。
- 严重副作用:角膜穿孔,过敏反应。
- 药物相互作用:未报告。
- 警告:在有腱病史的患者中谨慎使用。
- 广谱抗生素眼药水(例如,妥布霉素,庆大霉素):
- 费用:每瓶$10-$50。
- 禁忌症:对氨基糖苷类药物过敏。
- 副作用:眼部刺激,刺痛感。
- 严重副作用:角膜毒性,过敏反应。
- 药物相互作用:未报告。
- 警告:长期使用可能增加真菌或耐药细菌感染的风险。
- 抗病毒眼药水(例如,三氟尿苷,更昔洛韦):
- 费用:每瓶$50-$100。
- 禁忌症:对抗病毒剂过敏。
- 副作用:眼部刺激,刺痛感。
- 严重副作用:角膜毒性,过敏反应。
- 药物相互作用:未报告。
- 警告:在角膜完整性受损的患者中谨慎使用。
- 抗真菌眼药水(例如,纳他霉素,两性霉素B):
- 费用:每瓶$50-$100。
- 禁忌症:对抗真菌药物过敏。
- 副作用:眼部刺激,灼烧感。
- 严重副作用:角膜毒性,过敏反应。
- 药物相互作用:未报告。
- 警告:在角膜完整性受损的患者中谨慎使用。
- 糖皮质激素眼药水(例如,泼尼松龙,地塞米松):
- 费用:每瓶$10-$50。
- 禁忌症:现有的病毒或真菌感染。
- 副作用:眼内压升高,白内障形成。
- 严重副作用:角膜变薄,伤口愈合延迟。
- 药物相互作用:未报告。
- 警告:由于可能加重感染或延迟愈合,需在密切监督下谨慎使用。
手术:
- 角膜清创术:去除坏死组织和角膜溃疡中的碎屑以促进愈合。费用:$500-$1,500。
- 角膜贴片移植:在溃疡上放置捐赠者的角膜组织以促进愈合并恢复角膜完整性。费用:$2,000-$5,000。
- 羊膜移植:将羊膜应用于角膜溃疡以促进愈合和减少炎症。费用:$3,000-$6,000。
替代干预措施
- 自体血清眼药水:由患者自身血清制成的眼药水,含有生长因子和抗炎物质。费用:每瓶$100-$200。
- 绷带隐形眼镜:在角膜溃疡上放置治疗性隐形眼镜以保护角膜并促进愈合。费用:每片$100-$300。
- 羊膜提取物滴眼液:含有羊膜提取物的滴眼液,具有抗炎和促进伤口愈合的特性。费用:每瓶200-400美元。
- 富血小板血浆滴眼液:由患者自体富血小板血浆制成的滴眼液,富含促进愈合的生长因子。费用:每瓶300-500美元。
- 干细胞疗法:将干细胞注射到角膜溃疡中,以刺激组织再生和愈合。费用:每次治疗5,000-10,000美元。
生活方式干预
- 眼部卫生:指导患者保持良好的眼部卫生,在接触眼睛前洗手,并避免揉眼或抓挠受影响的眼睛。
- 隐形眼镜护理:教育患者正确的隐形眼镜护理,包括定期清洁、消毒和按照制造商建议更换。
- 眼部保护:建议患者在从事可能对眼睛造成伤害的活动时佩戴防护眼镜,如护目镜。
- 健康生活方式:鼓励患者保持健康的生活方式,包括均衡饮食、定期锻炼和充足睡眠,以支持整体眼部健康。
- 避免刺激物:建议患者避免接触烟雾、灰尘和化学品等刺激物,这些可能加重症状并延缓愈合。
需要注意的是,提供的费用范围是大致的,可能会因地点和干预措施的可获得性而有所不同。
Mirari 冷等离子体替代干预
了解Mirari 冷等离子体
- 安全和非侵入性治疗:Mirari 冷等离子体是一种安全和非侵入性的治疗选项,适用于多种皮肤状态。它不需要切口,从而减少瘢痕、出血或组织损伤的风险。
- 高效提取异物:Mirari 冷等离子体通过分解和剥离有机物,促进从皮肤移除异物,使得提取变得更容易。
- 减轻疼痛和舒适性:Mirari 冷等离子体具有局部镇痛效果,提供治疗期间的痛苦缓解,使患者更舒适。
- 感染风险减少:Mirari 冷等离子体具有抗菌特性,能够有效杀灭细菌并降低感染风险。
- 加速愈合和减少瘢痕:Mirari 冷等离子体刺激伤口愈合和组织再生,缩短愈合时间并减少瘢痕形成。
Mirari 冷等离子体处方
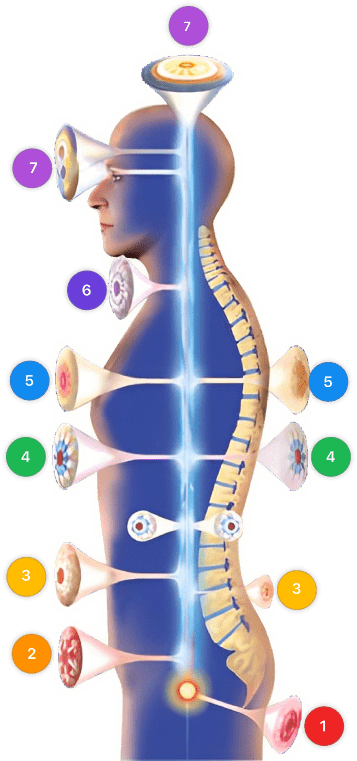
使用 Mirari 冷等离子体设备的视频说明 – F85 角膜溃疡 (ICD-10:H16.0)
| 轻度 | 中度 | 重度 |
| 模式设置:1 (感染) 位置:7 (神经系统 & 耳鼻喉) 早上: 15 分钟, 晚上: 15 分钟 |
模式设置:1 (感染) 位置:7 (神经系统 & 耳鼻喉) 早上: 30 分钟, 午餐: 30 分钟, 晚上: 30 分钟 |
模式设置:1 (感染) 位置:7 (神经系统 & 耳鼻喉) 早上: 30 分钟, 午餐: 30 分钟, 晚上: 30 分钟 |
| 模式 设置: 2 (伤口愈合) 位置: 7 (神经系统 & 耳鼻喉) 早上:15 分钟, 晚上:15 分钟 |
模式 设置: 2 (伤口愈合) 位置: 7 (神经系统 & 耳鼻喉) 早上:30 分钟, 午餐:30 分钟, 晚上:30 分钟 |
模式 设置: 2 (伤口愈合) 位置: 7 (神经系统 & 耳鼻喉) 早上:30 分钟, 午餐:30 分钟, 晚上:30 分钟 |
| 模式 设置: 3 (抗病毒治疗) 位置: 7 (神经系统 & 耳鼻喉) 早上:15 分钟, 晚上:15 分钟 |
模式 设置: 3 (抗病毒治疗) 位置: 7 (神经系统 & 耳鼻喉) 早上:30 分钟, 午餐:30 分钟, 晚上:30 分钟 |
模式 设置: 3 (抗病毒治疗) 位置: 7 (神经系统 & 耳鼻喉) 早上:30 分钟, 午餐:30 分钟, 晚上:30 分钟 |
| 总计 早晨: 45 分钟 大约 $7.50 美元, 晚上: 45 分钟 大约 $7.50 美元 |
总计 早晨: 90 分钟 大约 $15 美元, 午餐: 90 分钟 大约 $15 美元, 晚上: 90 分钟 大约 $15 美元, |
总计 早晨: 90 分钟 大约 $15 美元, 午餐: 90 分钟 大约 $15 美元, 晚上: 90 分钟 大约 $15 美元, |
| 通常 治疗周期 为 7-60 天 约为 $105 美元 – $900 美元 | 通常 治疗周期 为 6-8 周 约为 $1,890 美元 – $2,520 美元 |
通常 治疗周期 为 3-6 月 约为 $4,050 美元 – $8,100 美元
|
 |
|
使用Mirari冷等离子装置有效治疗角膜溃疡。
警告: MIRARI冷等离子是专为人体设计的,不包含任何人工或第三方产品。与MIRARI冷等离子结合使用其他产品可能会引起不可预测的效应、损害或伤害。在使用MIRARI前,请咨询医疗专业人士是否可以结合其他产品使用。
步骤1:清洁皮肤
- 首先使用温和的清洁剂或中性肥皂和水清洁皮肤受影响区域。用干净的毛巾轻轻拍干该区域。
步骤2:准备Mirari冷等离子装置
- 确保Mirari冷等离子装置已完全充电或根据制造商的要求装有新电池。确保装置干净且处于良好工作状态。
- 使用电源按钮或按照设备随附的具体说明打开Mirari装置。
- 一些Mirari设备可能有可调节的强度或治疗时间的设置。请遵循制造商的说明,根据您的需求和推荐的指南选择适当的设置。
步骤3:应用装置
- 将Mirari装置直接接触到皮肤受影响的区域。轻轻滑动或持有装置在皮肤表面上,确保均匀覆盖受影响区域。
- 慢慢地以圆周运动移动Mirari装置,或者按照用户手册中指示的特定模式操作,这有助于确保彻底的治疗覆盖。
步骤4:监测和评估:
- 跟踪您的康复进度,并评估Mirari装置在管理角膜溃疡方面的有效性。如果发现任何问题或不良反应,请咨询您的医疗保健专业人士。
注意
本指南仅用于信息目的,不应替代医疗专业人士的建议。始终咨询您的医疗保健提供者或合格的医疗专业人士以获取个人建议、诊断或治疗。不要仅依赖此处提供的信息来做出关于您健康的决定。使用此信息风险自担。该指南的作者及任何相关实体或平台不对基于内容的任何潜在不利影响或结果负责。
Mirari冷等离子系统免责声明
- 目的:Mirari冷等离子系统是一个二级医疗设备,旨在由受过训练的医疗专业人士使用。已注册在泰国和越南使用。不可用于这些地点之外的用途。
- 信息用途:随设备提供的内容和信息仅用于教育和信息目的,不是专业医疗建议或护理的替代品。
- 结果可变性:虽然设备已批准用于特定用途,但个人结果可能有所不同。我们不保证或断言具体的医疗结果。
- 咨询:在使用设备或根据其内容进行决策之前,重要的是咨询认证的Mirari远程治疗师和您的医疗保健提供者,了解具体协议。
- 责任:使用此设备即表示用户承认并接受所有潜在风险。制造商或供应商不对因使用设备而产生的任何不良反应、伤害或损害负责。
- 地理可用性:该设备已获得泰国和越南FDA的指定用途批准。到目前为止,除泰国和越南之外,Mirari冷等离子系统在其他地区不可购买或使用。
参考文献
- Eghrari AO, Riazuddin SA, Gottsch JD. 角膜综述:结构、功能和发展。Prog Mol Biol Transl Sci. 2015;134:7-23. doi:10.1016/bs.pmbts.2015.04.001
- 世界家庭医生组织 (WONCA). 国际初级保健分类,第二版 (ICPC-2). 牛津大学出版社; 1998.
- 世界卫生组织. 国际疾病和相关健康问题统计分类 (ICD-10). 世界卫生组织; 2019.
- Farahani M, Patel R, Dwarakanathan S. 感染性角膜溃疡。Dis Mon. 2017;63(2):33-37. doi:10.1016/j.disamonth.2016.09.005
- Austin A, Lietman T, Rose-Nussbaumer J. 感染性角膜炎管理进展。Ophthalmology. 2017;124(11):1678-1689. doi:10.1016/j.ophtha.2017.05.012
- Bourcier T, Thomas F, Borderie V, Chaumeil C, Laroche L. 细菌性角膜炎:风险因素、临床和微生物学综述。Br J Ophthalmol. 2003;87(7):834-838. doi:10.1136/bjo.87.7.834
- Tananuvat N, Punyakhum O, Ausayakhun S, Chaidaroon W. 泰国北部三级眼科中心的微生物角膜炎病因及临床结果。J Med Assoc Thai. 2012;95 Suppl 4:S8-S17.
- Dalmon C, Porco TC, Lietman TM, 等. 细菌性和真菌性角膜炎的临床区分:摄影调查。Invest Ophthalmol Vis Sci. 2012;53(4):1787-1791. doi:10.1167/iovs.11-8478
- Sharma S. 眼部感染性疾病的诊断。Eye (Lond). 2012;26(2):177-184. doi:10.1038/eye.2011.275
- Ung L, Bispo PJM, Shanbhag SS, Gilmore MS, Chodosh J. 微生物角膜炎的持续困境:全球负担、诊断和抗菌耐药性。Surv Ophthalmol. 2019;64(3):255-271. doi:10.1016/j.survophthal.2018.12.003
























